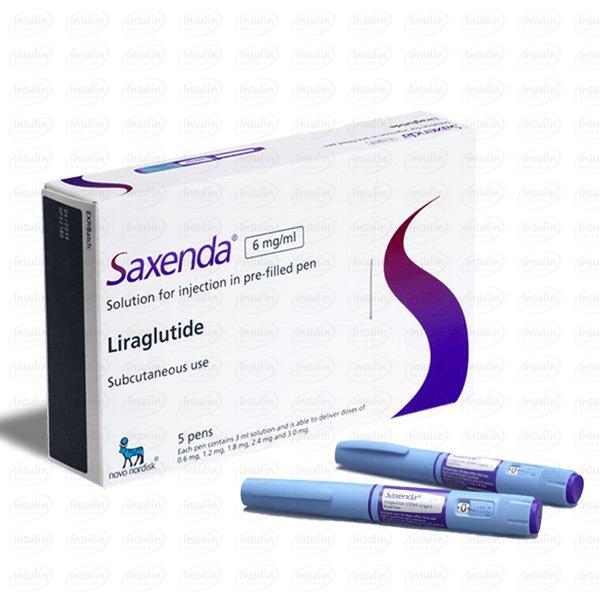
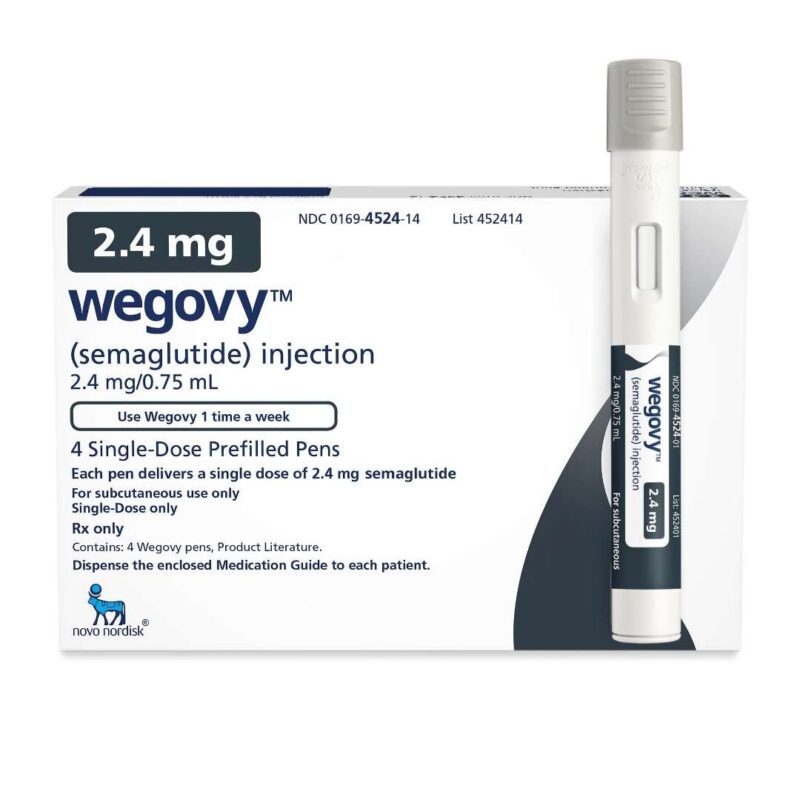
Saxenda (liraglutide) injection 3 mg is an injectable prescription medicine that may help some adults with excess weight (BMI ≥27) who also have weight-related medical problems or obesity (BMI ≥30) lose weight and keep the weight off. Saxenda should be used with a reduced-calorie meal plan and increased physical activity. Saxenda often gets confused with products that have similar functions such as Ozempic and Adlyxin. Saxenda should not be confused for any insulin product, such as pens, vials or cartridges. Similar to hormones found in the body, Saxenda regulates blood sugar, insulin levels, and digestion.
Tell your health care provider about all the medicines you take, including prescription, over-the-counter medicines, vitamins, herbal supplements and any kind of insulin (ie, isophane and degludec)
Saxenda slows stomach emptying and can affect medicines that need to pass through the stomach quickly. Saxenda is not known to be safe or effective in children under 12 years old. Saxenda is not known to be safe or effective in children with type 2 diabetes aged 12 to 17 years.
Saxenda may affect the way some medicines work and some other medicines may affect the way Saxenda works. Tell your health care provider if you take diabetes medicines, especially insulin and sulfonylurea medicines.
Saxenda is not for the treatment of type 2 diabetes nor should be combined with insulin. There is no specific diet with Saxenda but it would be beneficial to understand the foods to avoid.
| Saxenda | 1 Pen, 2 Pens, 3 Pens, 4 Pens, 5 Pens |
|---|



Reviews
There are no reviews yet.